See all "Itching" Section Topics

If you still have itching after a yeast infection treatment, it may be due to a few factors. You may be able to, via a self diagnosis, get a good idea as to what is causing your itch.
An important caveat is, even medical professionals have a difficult time diagnosing vaginal infections (i.e., yeast infections vs. bacterial vaginosis vs. trichomoniasis) without laboratory analysis (Botha, Van der Merwe; 2001).
Consequently, without a medical examination and microbiological assessment at a laboratory, it may be impossible to know definitely why you still have itching. However, through a little bit of understanding, you can possibly get a very good idea as to why your itching is still there after a yeast infection treatment was used.
So what are the primary reasons you are still itchy after you treated a yeast infection? The first possible reason is that the yeast infection is not actually gone.
Over the counter and prescription medications often do not resolve a Candida (Candida is the genus of fungi that causes all yeast infections; the term for a Candida infection is Candidiasis) infection.
This may be especially true with lower dose, over the counter preparations—such as Monistat®.
As research we will discuss demonstrates, a significant number of women who use over the counter and prescription antifungal medications do not see improvement of their situations.
And, a significant number of women only see improvement using a synthetic antifungal; and, symptoms appear to still persist.
The second possible reason you are still itchy after a yeast infection is treated is due to a secondary infection of bacterial vaginosis (BV). Often, many women have BV along with vaginal Candidiasis.
BV infections may cause itchiness, and they do not respond to antifungals—as they are caused by bacteria. Sometimes, BV causes symptoms that can help elucidate the etiology (cause) of vaginal problems.
BV is the most prevalent vaginal infection among women. BV is typified by vaginal dysbiosis (i.e., an imbalance in the microbial life in the vagina; a proliferation of unhealthy vaginal bacteria).
Dysbiosis occurs when pathogenic bacteria dominate the vaginal environment; and, these bacteria cause various symptoms.
Possibly the most well known symptom of BV is a foul "fishy" or "cheese" vaginal odor. BV also causes vaginal discharge; and, this discharge can be foul smelling as well. Importantly for this discussion, is the symptom of itching. BV can cause vaginal itching as well.
Because of the ubiquity of BV, this infection stands a decent chance to be the cause of your itching. it may be you have successfully treated your yeast infection; and it is gone.
Yet, BV is caused by bacteria, and will not be affected by antifungals. In fact, the overgrowth of Candida in the vagina may have upset the balance of "good" probiotic Lactobacilli (Lactobacillus is a genus of bacteria that normally dominates a healthy vagina).
If the Lactobacilli were displaced by an overgrowth of Candida, and the Candida subsequently wiped out, bad bacteria may have then taken advantage of the situation.
The abundance of space to propagate, and availability of resources, could have allowed pathogenic bacteria to dominate the vaginal microbiome (the environment of microscopic life). The result could then have been a BV infection that is causing itching.
A third possible reason you may still have itching is because you have the sexually transmitted disease (STD) trichomoniasis. This STD is caused by the protozoa Trichomonas vaginalis.
One symptom of this type of infection is itching. So, if you have had sexual activity that could put you in a situation to develop this STD; it is one possible culprit for your continued vaginal itching.
A caveat, if you just used a yeast infection medication, your infection may not be gone yet. Also, if the infection is gone, the damaged skin may need time to heal. It could be, if it has not been long since you took an antifungal of some kind, that itching is just due to damaged skin.
A yeast infection can damage skin slightly; thus, this healing skin could be itchy. This article focuses on itchiness that is occurring even after treatment has been completed for a few days.
So, if you used Monistat, and it is seven days or less since you used the treatment; itching could be due to damaged skin. In such instances, you will just need to be patient and use some itch reliever. Peppermint essential oil can relieve itchiness.
The following articles are linked below for your convenience; they may provide some answers you now are looking for. After this, we will discuss the research and get a better understanding of yeast infection persistence after treatment; and, explain BV in more detail.
- How Long Does it take for Monistat® to Work? (shows more cure rates for miconazole)
- Do I have a Yeast Infection or Bacterial Vaginosis?
- Yeast Infection and Sexually Transmitted Diseases
A 1/2 Day & Yeast is Gone!
Linda Allen suffered from yeast infections for years. Through researching natural medicine & Candida, she found an efficacious solution!
Linda is one expert you want on your side! Let her show you how to get rid of a superficial yeast infection in just 12 hours; AND, keep it gone!
A 60-day, 100% money back guarantee is provided.
Visit Official Site!Miconazole Cure Rate Percentages

Miconazole is the active ingredient in many Monistat® products. If you are using Monistat 1, 3, or 7; you are using the synthetic antifungal drug miconazole.
Therefore, the following studies may be very relevant; as they discuss how well miconazole works to alleviate yeast infections. As you will see, many women do not arrive at a complete cure using miconazole. This may be why you are still itchy after using Monistat.
Research, published in Acta Obstetricia Et Gynecologica Scandinavica [1990; 69 (5):417-422], evaluated how well a single 1,200mg intravaginal dose of miconazole worked to eliminate vaginal yeast infections. This research also evaluated fluconazole for a comparison.
The study encompassed a total of 99 women who had vaginal Candidiasis. The study excluded women with other vaginal infections, those who had recurrent vaginal Candidiasis (defined as more than 3 episodes in the last year), those who had used antifungal therapies, and those who were lactating or pregnant.
The healing ability of fluconazole and miconazole therapies were both evaluated with a score of ‘cure,’ ‘improvement,’ or ‘failure.’ The women participating in the study also evaluated these drugs with a score of ‘excellent,’ ‘good,’ ‘fair,’ or ‘not effective.’
In the study, 49 women took fluconazole, and 50 women took miconazole. The study stated that the two groups were comparable in all relevant respects (demographics, method of contraception, and dosing history). Six women in the group taking fluconazole were excluded from the efficacy evaluations for not complying with study protocols.
The study assessed the women at a short term follow up visit. Of those women taking miconazole, 39 (78%) were considered cured, 8 (16%) were considered to have improvement, and 3 (6%) were considered to have failed attaining a cure.
Concerning the long term follow up examination; miconazole effected a cure in 40 (80%) of the women, improvement in 3 (6%) of the women, and the drug failed in 7 (17%) of the women. The chart below, taken from the study, shows graphically these findings for miconazole and also states the findings for fluconazole.
![The above chart was taken from a study published in Acta Obstetricia Et Gynecologica Scandinavica [1990; 69 (5):417-422]. The chart shows how 1200mg intravaginal dose of miconazole (the active ingredient in Monistat®) and fluconazole worked at getting rid of vaginal yeast infections. As the chart indicates, a significant number of women did not see full improvement using the intravaginal dose of miconazole.](/img/How-Miconazole-and-Fluconazole-Cure-Yeast-Infections.png)
Concerning the women’s self assessment (these women’s own opinions of the treatment), there were a significant number of women who evaluated the miconazole treatment as not effective—at both short and long term evaluations.
For the short term self assessment, 26 (52%) women reported the miconazole treatment as excellent, 16 (32%) of the women reported it as good, 4 (8%) women reported it to be fair, and 4 (8%) reported it as not effective.
For the women’s long term self assessment, 28 (56%) women reported the miconazole treatment as excellent, 10 (20%) reported it as good, 3 (6%) reported it as fair, and 9 (19%) reported it as not effective. The chart below was taken from the study, and shows these statistics (also included are the responses for the fluconazole therapy).
![The above chart shows how women in a study (published in Acta Obstetricia Et Gynecologica Scandinavica [1990; 69 (5):417-422]) evaluated the performance of fluconazole (brand name Diflucan) and miconazole (the active ingredient in Monistat®). As the chart indicates, there are a significant number of women who reported that miconazole did not perform well in eliminating their vaginal yeast infections.](/img/Womens-Opinions-of-Miconazole-for-Vaginal-Candidiasis.png)
Concerning the evidence from microbial cultures (where samples of the women’s vaginal material were grown to see if Candida cells were present), all of the women in the study had a positive microbial culture at the start of the study.
At the short term follow up, only 1 women taking fluconazole and 2 women taking miconazole tested positive for having Candida. Oddly enough, none of these 3 women who tested positive for Candida had any signs or symptoms of vaginal yeast infections.
Long term laboratory cultures showed that 26% of the women taking fluconazole had Candida. Long term laboratory cultures also showed that 82% of the women who used miconazole had Candida in their vaginas.
This therefore indicates the propensity of Candida to reinvade the vagina. Although, the mere presence of Candida is common; infection is not guaranteed by the mere presence of this yeast.
Another study looked at the ability of miconazole to treat severe vaginal Candidiasis. This research was published in Gynecologic and Obstetric Investigation [80.2 (2015): 113-118]. According to the study’s authors, miconazole is a imidazole (a type of azole drug) antifungal which possesses a wide range of ability against C. albicans and other species of Candida.
The goal of the study was to evaluate the ability of a miconazole vaginal suppository and oral fluconazole for their ability to treat severe vaginal Candidiasis.
The study used a particular way to differentiate severe vaginal Candidiasis from normal vaginal Candidiasis. For this purpose, the severity of signs and symptoms were assessed. Symptoms such as itching, erythema (the reddening of the skin; often in patches), burning, and vaginal discharge were taken into consideration.
The symptoms were given a score corresponding to their severity: 0 = absent, 1 = mild, 2 = moderate, and 3 = severe. Those women who gave an overall severity score for symptoms of 7 or more were considered to have severe vaginal Candidiasis.
The women who were utilized by this research were generally healthy and had severe vaginal Candidiasis. 577 women completed this study; and, they were 19 to 45 years old. 320 women used intravaginal miconazole, and 30 women in this group dropped out of the study. 320 women took fluconazole and 33 in this group dropped out of the study.
Women with recurrent vaginal Candidiasis were excluded from the study. During the study, women agreed to not use any other vaginal product. Women were also excluded from this research if they had a disease known to predispose to yeast infections (such as diabetes), or were taking antibiotics or corticosteroids.
Additionally, pregnant women, and those who used an antifungal medication in the week before study entry, were not allowed to participate in the research.
The medication the women in the study used was either miconazole or fluconazole. The women who used miconazole took a 1,200mg vaginal suppository at bedtime on days 1 and 4. Those women taking fluconazole used an oral dose of 150mg on day 1 and another 150mg dose on day 4.
After taking the last dose of their respective medications, the women were required to have a follow-up visit 7 to 14 days later. A second follow up visit 30 to 35 days after taking the last dose of medication was also required. At these visits, the signs and symptoms of infection were scored and compared to the original assessment before receiving treatment.
The study defined a clinical cure as the general absence of signs and symptoms with a total severity score of 2 or less. Improvement was defined as a considerable reduction of the severity of signs and symptoms accompanied by a decrease in the severity score of 50% or more. Those women who did not improve, or arrive at a clinical cure, were considered to be treatment failures.
The results for the women using the intravaginal miconazole were as follows at the first follow-up visit (day 14):
- Clinical cure was seen in 288 (78.6%) women.
- Improvement was seen in 28 (9.6%) women.
- Treatment failure was seen in 34 (11.7%) women.
The results for the women using the intravaginal miconazole were as follows at the second follow-up visit (day 35):
- Clinical cure was seen in 235 (81%) women.
- Improvement was seen in 13 (4.4%) women.
- Treatment failure was seen in 42 (14.4%) women.
As we can see from this study, the women used two 1,200mg doses of miconazole intravaginally on days 1 and 4. According to Monistat’s website, Monistat 1 Day contains a 1,200mg dose of miconazole.
Therefore, this study is generally going to show the results for using two Monistat 1 Day products 3 days apart. As you can tell, there are going to be a significant number of women who do not arrive at a clinical cure using Monistat 1 Day.
It may be that a single application of 1,200mg miconazole will be even less effective than two 1,200mg doses of this drug.
Consequently, if you already used Monistat, it may be that you still have itching after a yeast infection treatment due to the infection not being totally gone.
Eliminate Bacterial Vaginosis & Vaginal Odor
Jennifer O’Brien is one prominent expert on BV that knows how to get rid of vaginal odor. BV is a common infection that you don’t have to put up with.
Jennifer will show you how to naturally eliminate vaginal odor in just 3 days.
A 60-day, 100% money back guarantee is provided.
Visit Official Site!Fluconazole (Diflucan) Cure Rate Percentages
For this discussion, it may be important to know that Diflucan is a common brand name for the drug fluconazole.
A study, published in Drugs [39.6 (1990): 877-916], examined research relating to fluconazole. This research shows clearly that fluconazole does not totally cure a significant minority of women with vaginal Candidiasis.
According to this review, the results from a large trial revealed that of 146 women with symptomatic vaginal Candidiasis (i.e., the infection had generated symptoms) 134 (92%) of these women were clinically cured (this may mean that signs and symptoms had gone away; yet, there may have still been Candida living in the vagina) by a single oral dose of 150mg of fluconazole.
Yet, in a longer term evaluation of these women, at 27 to 62 days after taking the medication, 83% of these women were clinically cured, 5% had improved, and 12% of the women had no improvement (i.e., they had a vaginal yeast infection still, or it had returned).
The review also reports that 10% of the women who were initially clinically cured had redeveloped yeast infection symptoms.
For the laboratory analysis, the Candida cells were totally killed in 94% of the women in the short term evaluation. Concerning the long term evaluation, 73% of the women remained free of Candida cells in their vagina.
This review also cites another study that showed the rate of cure fluconazole would produce. This cited research was done on women with normal, not chronic, yeast infections.
This cited study showed that fluconazole totally eliminated Candida cells from these women’s vaginas in 80% of the women 7 to 28 days after they took a single oral dose of 150mg of fluconazole.
This review also cites another study that compared fluconazole with ketoconazole (ketoconazole is another azole antifungal drug). A 150mg single oral dose of fluconazole was compared to an oral dose of 200mg ketoconazole which was taken twice a day for 5 days.
This cited research utilized 183 women who had vaginal Candidiasis.
This cited study found a favorable reduction of symptoms was attained in the short term evaluation by both drug regimens. At another long term evaluation, 27 to 62 days later, favorable resolution of symptoms was evident in 86% of the women who used fluconazole and 88% of the women who used the 5 day ketoconazole treatment.
Yet, only 77% of the women treated with either drug were found to have Candida completely eradicated from their vaginas.
Fluconazole & Yeast Infection Recurrence
Another study discusses the ability of fluconazole to suppress the occurrence of yeast infections. The study was published in Expert Review of Anti-Infective Therapy [2.3 (2004): 357-366]. Recurrent vaginal Candidiasis plagues many women; and, they often do not find a solution using conventional drugs.
The authors of this study stated that cures are difficult to attain for women with recurrent vaginal Candidiasis. Yet, the study continues to say that suppressive maintenance therapy (using fluconazole even when a yeast infection is not present in an attempt to keep them from happening) with oral fluconazole is highly effective in managing symptoms.
This study cites research that found a weekly maintenance therapy, of orally taking 150mg of fluconazole, protected over 93% of the women from having another yeast infection.
The study goes on to remark that suppressive maintenance therapy with an antifungal drug can provide a better quality of life to women with recurrent vaginal yeast infections. And, the study also states that recurrent infections can happen, at times, on a monthly basis.
Yet, the downside to repressive maintenance therapy is a high rate of relapse. The study states that after a 6 month course of suppressive antifungal therapy is finished, approximately 40% of the women will relapse—developing further vaginal yeast infections. The study states that suppressive therapy with fluconazole has been shown to be safe and well tolerated.
Clair Goodall: Author & Nature Lover
Clair Goodall is a bee-obsessed natural medicine convert from Minnesota. She is one expert you might want to know more about!
Clair will help you protect you and your family from toxic products and chemicals and help you discover solutions from nature.
Also, Clair’s book is backed by a 60-day, 100% money back guarantee
Visit Official Site!Bacterial Vaginosis: Another Instigator of Vaginal Itching
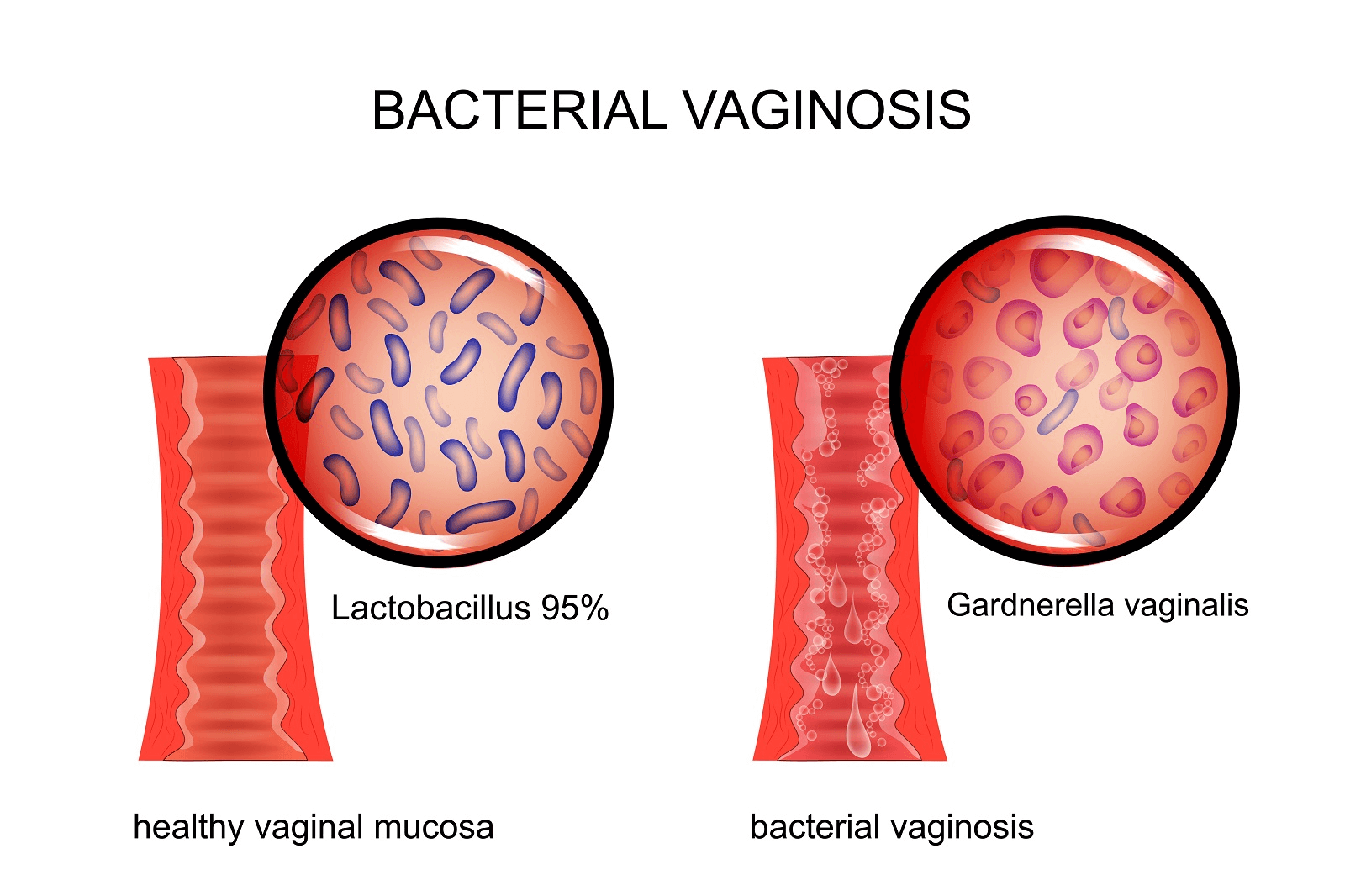
A study discussing BV was published in BJOG: An International Journal of Obstetrics & Gynaecology [124.1 (2017): 61-69]. According to the study, BV is the most prevalent lower genital tract disease in women of reproductive age. The study states that an important characteristic of BV is a change in the microbiological life of the vagina.
A healthy woman’s vagina is going to be dominated by Lactobacillus bacteria (you may be familiar with the popular probiotic Lactobacillus acidophilus). In BV, the Lactobacillus dominated vaginal microbiota (the microscopic organisms living in an area) changes.
Lactobacilli no longer dominate the microbiome (the environment of microscopic life); instead, other anaerobic and facultative (i.e., the bacteria can live with or without oxygen) bacteria do.
Concerning itching, BV is known to cause vaginal itching. The following studies show the prevalence of itching as a symptom of BV:
- A study in Journal of Advanced Pharmaceutical Technology & Research [9.1 (2018): 15] reports that itching occurred in 10 (12.5%) out of 80 women with bv.
- A study in the Annals of Internal Medicine [2008 149:1, I-30] reported that women with BV often do not have symptoms. Some women with BV will have vaginal itching. Other symptoms that can occur with BV include vaginal odor and discharge.
- An article provided by the United States National Center for Biotechnology Information (click here to view the article), written by Norah Kairys and Manish Garg, stated that vaginal itching can be a symptom of BV. The article states that most women with BV will have a foul smelling vaginal discharge. This foul smelling discharge can be more noticeable after sexual intercourse. Additionally, it is stated that many women with BV can be without symptoms.
According to a study published in the Journal of the American Academy of Physician Assistants [30.12 (2017): 15-21], BV is the most common cause of vaginal odor and vaginal discharge. Another symptom BV can cause is vaginal irritation.
Concerning the prevalence, the research states BV affects about 29% of all women. This study states that BV often recurs after treatment; with 50% of women getting the infection again within 12 months. And, BV predisposes women more to acquiring a sexually transmitted disease, including HIV.
The study also states that the "gold standard" for diagnosing BV is through Gram staining and Nugent scoring. This method involves identifying and quantifying Lactobacillus bacteria in addition to other bacteria (such as Gardnerella vaginalis, Bacteroides, Mobiluncus). An overgrowth of Gardnerella vaginallis in the vagina is one indicator of a woman having BV.
A 100% Natural, 12 Hour Yeast Infection Remedy

Yeast infections in general can be an arduous experience. No one wants to have this problem. Candida can attack both men and women; this is also true for more systemic infections and gut yeast infections.
Yet, women appear to be more prone to this infection’s recurrence; and, this condition can negatively impact a woman’s quality of life in a very significant way.
One woman who suffered from recurrent yeast infections was Linda Allen. Linda’s health problems began in her late teen years. Eventually Linda developed a yeast infection. After visiting a doctor about her infection, she was prescribed medication. The medication she was given by the doctor solved her problem.
However, it was not long before Linda developed another yeast infection. This cycle of yeast infection recurrence and treatment would go on for a long time. In all, Linda would spend about 12 years dealing with recurrent yeast infections.
During these 12 years, yeast infections were not Linda’s only health problem. She developed a variety of symptoms that appeared to have no understandable cause. Her doctor did not know what was causing her terrible health either; they even tried putting Linda on allergy medication.
Nothing seemed to work, and Linda’s health was simply terrible. It appeared that there was no recourse for her terrible, strange illness. Linda and her doctor, at this time, did not know that Linda was suffering from systemic Candidiasis. Candida had overgrown in her body and was deteriorating her health.
The situation of Linda Allen’s life changed one day for the better. This amazing event happened when Linda decided to get the council of a naturopathic practitioner. The naturopath knew exactly what was causing Linda’s poor health and recurrent yeast infections: systemic Candidiasis.
Although the naturopath did not provide Linda with a permanent solution to end her Candida problem, they did point her in the right direction.
From this time on, Linda began to devour books and information about Candida and natural medicine. Linda also inquired of health professionals who were kind enough to lend her some of their time.
Eventually, Linda developed a natural system for eliminating the Candida from her body. Before she tried it on herself, she spent a year refining this new approach. When Linda put her new system into practice, she began to experience amazing results.
The recurrent yeast infections that had plagued Linda’s life suddenly stopped. As time went on, her entire health began to remarkably improve. Eventually, she had cleansed her body of Candida. The result of curing her systemic yeast infection was a resolution of her symptoms and the restoration of her full health. Linda had done it!
After her success, and the success of others she gave her new system to, Linda went on to write an entire book about her system. Linda’s book contains her natural system that can totally wipe out systemic yeast infections.
And, for those who are only suffering from a "regular" yeast infection, she provides a natural system for eliminating them in 12 hours of time. Through using Linda’s system, you can avoid harsh synthetic drugs and be free from the symptoms of a yeast infection in about half a day.
Linda has published her book with a large online retailer; a subsidiary of the United States based firm Keynetics Incorporated. Linda’s publisher handles the distribution of her book and offers a 60 day, 100% money back guarantee on Linda Allen’s book.
In addition to protecting her clientele with a wonderful refund guarantee, her publisher also makes the purchasing process secure and easy. If you need support, Linda’s publisher can provide the assistance you need. Linda’s publisher does excellent work.
If you would like to learn more about Linda’s personal journey to recovery, see testimonials of others who tried her book, or find out more about Linda’s book; you can learn more at Linda Allen’s website.
Since Linda Allen published her book, she has helped over 100,000 people from around the world become free of Candida problems. Linda is truly a great expert to consult when it comes to Candida related health problems!
Author: Mr. Nicholas Gross

Nick Gross is a natural medicine enthusiast who has been researching and writing about natural medicine since 2008. Nick is primarily a web developer but also researches and authors written and video content about natural health. Nick has a bachelor’s degree in Management Information Systems from the University of Northern Iowa.
Disclaimer
The information on this website is not a prescription for anyone. This information is for informational or educational purposes only, and is not a substitute for professional medical advice or consultations with healthcare professionals.
Affiliate Disclosure
Some of the links provided on this website are affiliate links. When a purchase is made through these links, Candida Hub earns money from commission. This helps to keep the website up and helpful to people for free. Thank you for any support!
Stay Up to Date
If you enjoyed this article, consider following / liking our Facebook page. This page is primarily utilized to alert followers of new articles that are put on Candida Hub. Candida related news is also discussed. While you are there, you can see what has been more recently added to Candida Hub.
SOURCES:
- https://doi.org/10.4102/hsag.v6i1.54 — Botha, D. E., and R. Van der Merwe. "Diagnosis of vaginal infection in pregnancy: research." Health SA Gesondheid [6.1 (2001): 21-27].
- https://doi.org/10.3109/00016349009013305 — van Heusden AM, Merkus HM, Corbeij RS, et al. “Single-dose oral fluconazole versus single-dose topical miconazole for the treatment of acute vulvovaginal candidosis.” Acta Obstetricia Et Gynecologica Scandinavica [1990; 69 (5):417-422].
- https://doi.org/10.1159/000371759 — Fan, Shangrong, Xiaopingliu Liu, and Yiheng Liang. "Miconazole nitrate vaginal suppository 1,200 mg versus oral fluconazole 150 mg in treating severe vulvovaginal candidiasis." Gynecologic and Obstetric Investigation [80.2 (2015): 113-118].
- https://doi.org/10.2165/00003495-199039060-00006 — Grant, Susan M., and Stephen P. Clissold. "Fluconazole." Drugs [39.6 (1990): 877-916].
- https://doi.org/10.1586/14787210.2.3.357 — Cha, Raymond, and Jack D. Sobel. "Fluconazole for the treatment of candidiasis: 15 years experience." Expert Review of Anti-Infective Therapy [2.3 (2004): 357-366].
- https://doi.org/10.1111/1471-0528.14209 — Nasioudis, Dimitrios, et al. "Bacterial vaginosis: a critical analysis of current knowledge." BJOG: An International Journal of Obstetrics & Gynaecology [124.1 (2017): 61-69].
- https://doi.org/10.4103/japtr.JAPTR_305_17 — Pazhohideh, Zahra, et al. "The effect of Calendula officinalis versus metronidazole on bacterial vaginosis in women: A double-blind randomized controlled trial." Journal of Advanced Pharmaceutical Technology & Research [9.1 (2018): 15].
- https://doi.org/10.7326/0003-4819-149-1-200807010-00002 — Thomas, T. L., K. Ringwood, and D. N. Fredricks. "Factors Related to Bacterial Vaginosis That Persists or Occurs Again after Treatment in Women Who Have Sex with Women." Annals of Internal Medicine [2008 149:1, I-30].
- https://www.ncbi.nlm.nih.gov/books/NBK459216/ — Norah Kairys, Manish Garg. “Bacterial Vaginosis.” StatPearls Publishing LLC [2019]
- https://doi.org/10.1097/01.JAA.0000526770.60197.fa — Bagnall, Paulette, and Denise Rizzolo. "Bacterial vaginosis: a practical review." Journal of the American Academy of Physician Assistants [30.12 (2017): 15-21].







