See all "Gut Yeast" Section Topics
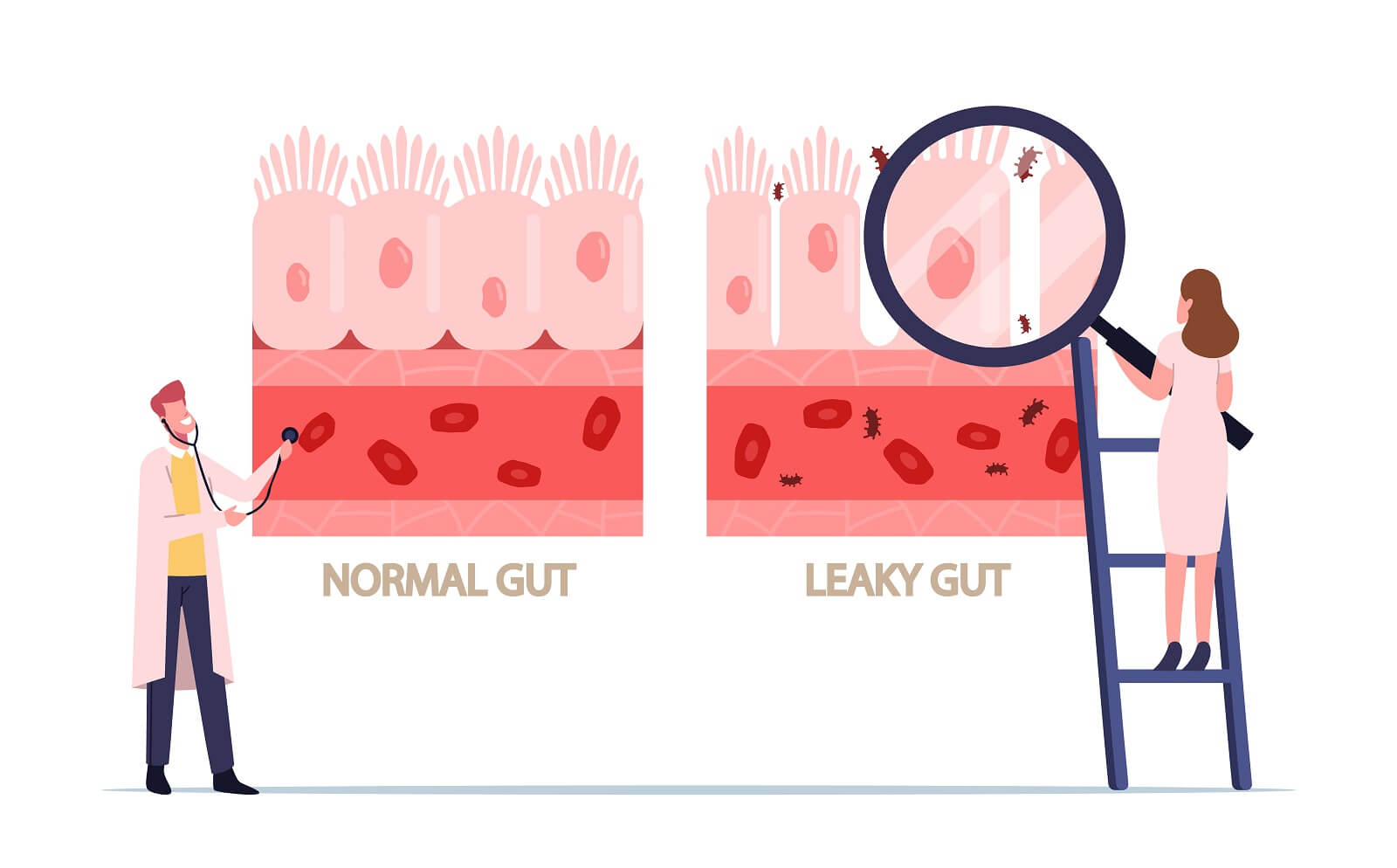
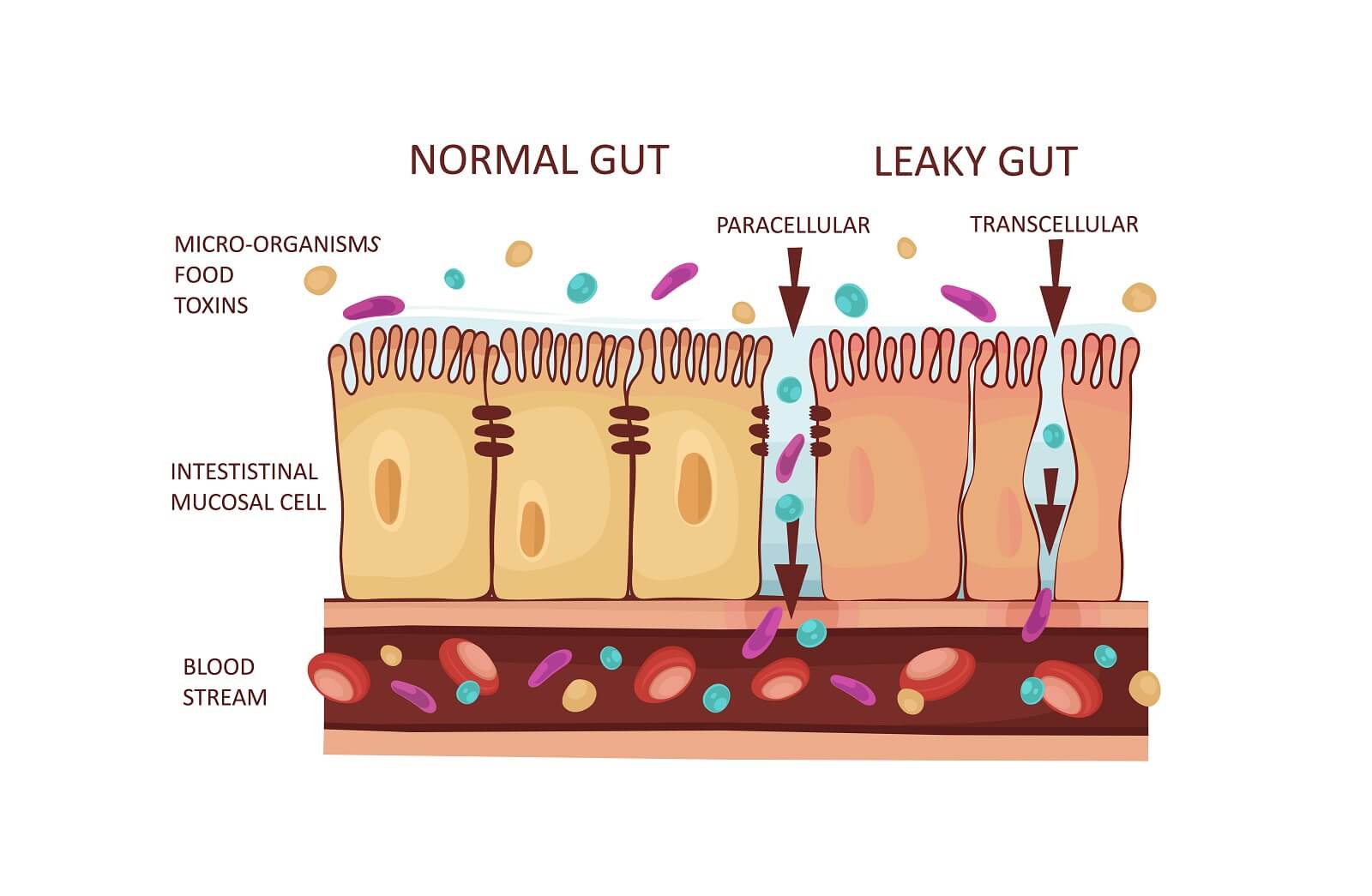
Candida species that grow hyphae (long, thin germ tubes), may be a factor in causing, or aggravating, leaky gut syndrome. The hyphal growths, of various Candida species, have been proven to elevate their virulence and ability to invade other sectors of the body. These hyphae, can burrow into the walls of the intestines. Additionally, Candida also secretes an enzyme that breaks down the protective layers of mucous that typically form a chemical barrier to the food and microbiota traveling through the intestines. By attaching itself to the mucous membranes of the intestines, dissolving the protective mucous, and drilling holes into the intestinal wall, Candida may instigate leaky gut syndrome; or aggravate this condition.
The toxins Candida yeast create, also may weaken the immune system. This speculation, was made by a study published in Contributions to Microbiology and Immunology [4 (1976): 77-85]. With the physical progress yeast can make through the gut into the bloodstream, and the potential stymying of the immune system via toxins, Candida may be a significant detriment to health. Other things, such as partially digested food and microorganisms, may also follow through the breaches Candida makes in the intestinal wall. This could, in turn, result in various allergic reactions and bacterial infections throughout the body.
Inflammation and Leaky Gut Syndrome
Inflammation of the intestinal walls can cause the tiny pores, that normally allow digested nutrients into the bloodstream, to widen. Once these pores become too big; food, bacteria, and yeast can start to slip through and enter the bloodstream. According to a study published in Current Opinion in Microbiology [14.4 (2011): 386-391], inflammation increases Candida overgrowth; and, Candida overgrowth worsens inflammation. According to the study:
Since inflammation increases the likelihood of significant Candida colonization and Candida colonization reduces healing of lesions, these effects would produce a vicious cycle. The presence of inflammation alters bacterial colonization and the activities of the host, creating conditions that favor both high level Candida colonization and exacerbation of disease.
Current Opinion in Microbiology [14.4 (2011): 386-391]
Consequently, Candida can start a terrible cycle into motion in the digestive system: prompting inflammation and spreading to the newly inflamed areas. The only way to effectively stop this, is to eliminate the overgrowth of yeast in the gut. This should reduce the size of the pores in the intestines, and help to ameliorate leaky gut syndrome.
A 1/2 Day & Yeast is Gone!
Linda Allen suffered from yeast infections for years. Through researching natural medicine & Candida, she found an efficacious solution!
Linda is one expert you want on your side! Let her show you how to get rid of a superficial yeast infection in just 12 hours; AND, keep it gone!
A 60-day, 100% money back guarantee is provided.
Visit Official Site!Candida Degradates Intestinal Surfaces
A study, published in Infection and Immunity [64.11 (1996): 4514-4519], found that Candida albicans was able to break down the mucin produced by the intestinal mucous membranes with a proteinase enzyme (an enzyme that can break down certain proteins). The researchers speculated, that this was a key element in how Candida albicans was able to break through the chemical barrier provided by mucins surrounding the intestine’s epithelial walls. The researchers in the study, cited various research, that proved Candida albicans secretes an enzyme which breaks down proteins and can degredate epithelial keratin (a protein which protects epithelial cells), dermal collagen (the intestines contain collagen as well), albumin (the main protein in human blood), hemoglobin (a protein which carries oxygen), and immunoglobulin A (an antibody that plays a critical role in mucosal immunity).
Another study, published in FEMS Microbiology Letters [153.2 (1997): 349-355], showed that Candida albicans was able to break down the endothelium. The endothelium is a thin layer of flat epithelial cells (cells which cover, or line, various body surfaces) that lines the lymph vessels, blood vessels, and the inner cavities of the heart. Thus, given Candida’s ability to break down this layer of cells, it could enter through the epithelium (a layer of cells surrounding the outer part of the intestinal wall), break down the endothelium, and enter into the bloodstream. The researchers stated the following:
Candida albicans infections in severely immunocompromised patients are not confined to mucosal surfaces; instead the fungus can invade through epithelial and endothelial layers into the bloodstream and spread to other organs, causing disseminated infections with often fatal outcome… Our results demonstrate that the C. albicans acid proteinase degrades human subendothelial extracellular matrix; this may be of importance in the penetration of C. albicans into circulation and deep organs.
FEMS Microbiology Letters [153.2 (1997): 349-355]
Another study, conducted on mice by giving them intestinal yeast infections, was published in Medical Mycology [42.5 (2004): 439-447]. The study used two primary types of Candida albicans: strains that could produce normal hyphal growth, and strains that failed to produce normal hyphal growth. The study found that the strains that could produce germ tube hyphae, were much more virulent. The strains capable of normally forming hyphae, would kill the mice they were tested on; and, could travel from the intestines to other organs in the mice. Some of the mice, with the virulent hyphal strains of Candida albicans, died from Candida colonizing their kidneys. The reduced presence, of hyphal growth deficient yeast, in examined mice body tissue and lack of colonization efficacy; was suggested by the researchers, to potentially be due to the lack of adequate hyphal growth. Hyphal growth deficient yeast, were also unable to kill the mice after they were inoculated. Consequently, this study proves that, Candida albicans, can move from the gut to other areas of the body; likely due to its ability to burrow into the walls of the intestines with hyphal growths.
Eliminate Bacterial Vaginosis & Vaginal Odor
Jennifer O’Brien is one prominent expert on BV that knows how to get rid of vaginal odor. BV is a common infection that you don’t have to put up with.
Jennifer will show you how to naturally eliminate vaginal odor in just 3 days.
A 60-day, 100% money back guarantee is provided.
Visit Official Site!Hyphal Candida Species
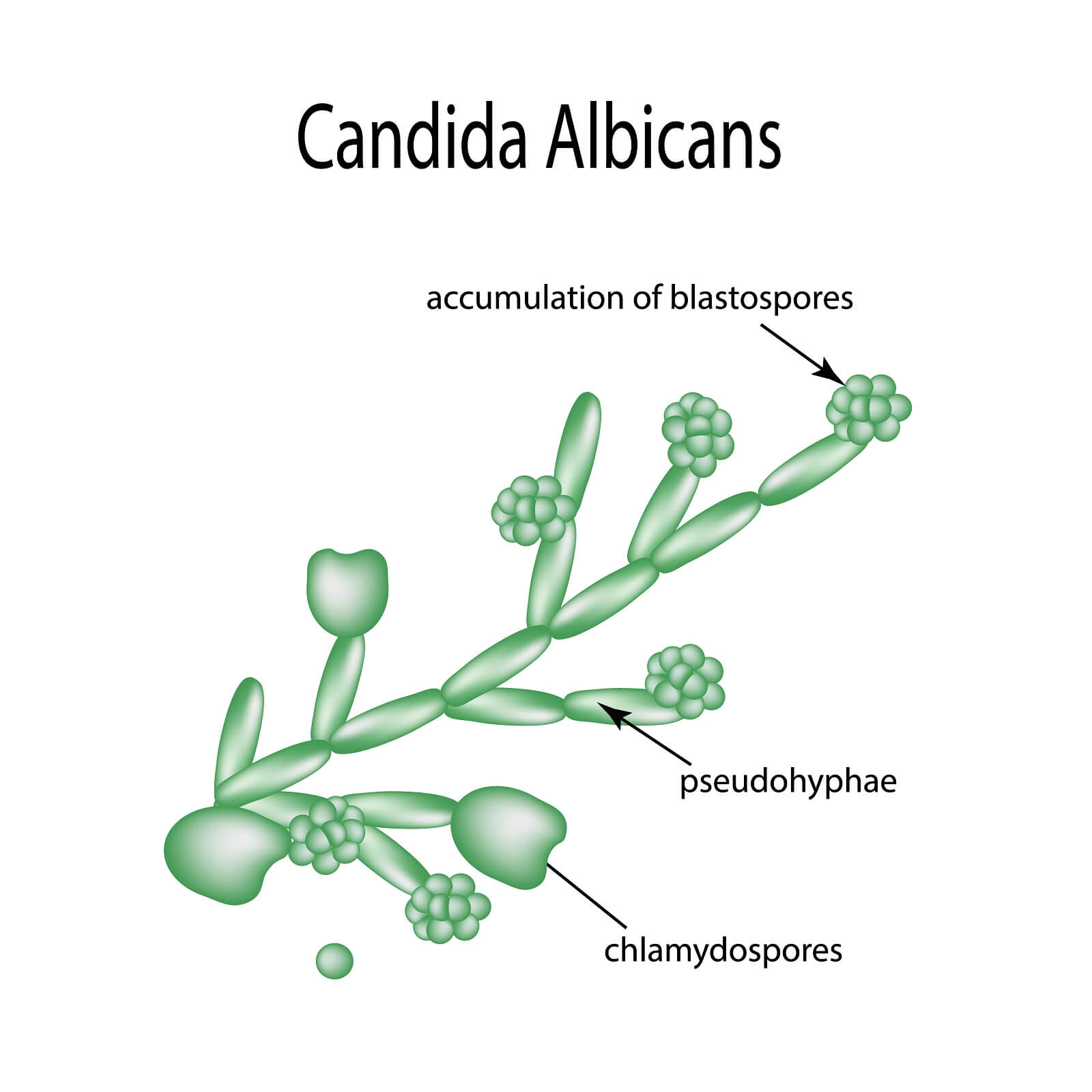
Not all species of Candida grow hyphae. Some are dimorphic, meaning they exist as single cells (blastoconidia) and as germ tubes (hyphae). Before 1995, all germ tube positive Candida species were identified as Candida albicans. According to a study published in Indian Journal of Pathology and Microbiology [40 (1997): 55-58], Candida albicans and Candida parapsilosis show very strong hyphal growth. For more information on Candida’s hyphal growth, feel free to check out this article: Candida Hyphae Germ Tubes. The following species form hyphae, and are more virulent than Candida species that do not form hyphae:
- Candida albicans
- Candida tropicalis
- Candida parapsilosis
- Candida dubliniensis
Clair Goodall: Author & Nature Lover
Clair Goodall is a bee-obsessed natural medicine convert from Minnesota. She is one expert you might want to know more about!
Clair will help you protect you and your family from toxic products and chemicals and help you discover solutions from nature.
Also, Clair’s book is backed by a 60-day, 100% money back guarantee
Visit Official Site!A Natural, 12 Hour Yeast Infection Cure

According to a research paper published in Clinical Microbiology Reviews [12.1 (1999): 80-96], Candida species are quite ubiquitous organisms. Candida are most frequently present in the mouth; and, live in 31% to 55% of healthy people. The species that causes approximately 70% to 80% of all Candida infections is C. albicans.
The Chinese Journal of Obstetrics and Gynecology [2011 Jul;46(7):496] reports there appears to be a correlation between intestinal Candida infections and vaginal yeast infections. And, this provides a clue, as to why yeast infections in general, can reoccur.
This study states, in 148 cases of vaginal candida infections, 33.1% of the women were infected in both the intestines and vaginal area. The recurrence rate of yeast infections, in women with simultaneous intestinal infection, was significantly higher than for women who did not have an intestinal infection. This study concluded that vaginal yeast infections are highly associated with simultaneous intestinal Candida infection.
As research appears to indicate, systemic Candida infections can and do happen. A more systemic Candida infection may primarily get a foothold in the intestines; and cause a wide array of problems. If your yeast infections keep happening, a systemic Candida problem may be why.
One woman who suffered from a systemic Candida infection, for about 12 years, was Linda Allen. The systemic Candida infection that attacked Linda caused a wide range of health problems in addition to yeast infections. Some of these problems, Linda describes in her own words in the following quote:
To be honest, it was hard to pinpoint exactly what was wrong: I wasn’t really sick, but I wasn’t really well either. I had listlessness, fatigue, brain fog, stomach ailments, unexplained rashes, skin infections, and so on. It seemed like every day brought a new challenge.
My energy was sapped and I felt exhausted, which affected my grades and put a big dent in my social life.
Linda Allen’s symptoms included an embarrassing vaginal discharge, severe itching, and burning sensations. Her infections were difficult to deal with, and Linda’s health problems cost her financially as well. Linda states these infections of Candida can become excruciating when they happen as frequently as a menstrual period.
Yet, Linda spent a great deal of time in research; and even questioned health professionals who were kind enough to share some time with her. Linda even tried an array of purported "cures." Although it took a while, eventually, Linda put together a natural treatment plan she hoped would solve her Candida situation.
After spending about a year refining her new approach, Linda tried her system on herself. It worked amazingly well. Linda even returned to a few medical doctors to get tested for the presence of infections. These tests revealed all indicators of infection had vanished! Linda was indeed well again, after such a long, difficult journey.
Linda has since published a book detailing how to copy her success. She also includes a 12 hour yeast infection cure that can get rid of a superficial (such as a genital yeast infection or oral thrush) yeast infection in about 12 hours.
Linda’s publisher protects those who get her book with a 60 day, 100% money back guarantee. Linda’s publisher, a subsidiary of the United States based firm Keynetics Incorporated, is a reputable digital retailer that has been around for a long time. They have great customer service, and make getting a full refund on Linda’s book quick and easy. If you’re not satisfied, you can quickly get all your money back.
If you would like to learn more about Linda’s journey to freedom from Candida, see reviews of others who tried her natural system, or find out more about her efficacious book; you can find more information at Linda Allen’s website.
Author: Mr. Nicholas Gross

Nick Gross is a natural medicine enthusiast who has been researching and writing about natural medicine since 2008. Nick is primarily a web developer but also researches and authors written and video content about natural health. Nick has a bachelor’s degree in Management Information Systems from the University of Northern Iowa.
Disclaimer
The information on this website is not a prescription for anyone. This information is for informational or educational purposes only, and is not a substitute for professional medical advice or consultations with healthcare professionals.
Affiliate Disclosure
Some of the links provided on this website are affiliate links. When a purchase is made through these links, Candida Hub earns money from commission. This helps to keep the website up and helpful to people for free. Thank you for any support!
Stay Up to Date
If you enjoyed this article, consider following / liking our Facebook page. This page is primarily utilized to alert followers of new articles that are put on Candida Hub. Candida related news is also discussed. While you are there, you can see what has been more recently added to Candida Hub.
SOURCES:
- http://www.ncbi.nlm.nih.gov/pubmed/342194 — Iwata, K. "Toxins produced by Candida albicans." Contributions to microbiology and immunology 4 (1976): 77-85.
- http://dx.doi.org/10.1016%2Fj.mib.2011.07.015 — Kumamoto, Carol A. "Inflammation and gastrointestinal Candida colonization." Current opinion in microbiology 14.4 (2011): 386-391.
- http://www.ncbi.nlm.nih.gov/pmc/articles/PMC174406/ — Colina, Ana-Rosa, et al. "Evidence for degradation of gastrointestinal mucin by Candida albicans secretory aspartyl proteinase." Infection and Immunity 64.11 (1996): 4514-4519. PDF Available Here
- http://dx.doi.org/10.1111/j.1574-6968.1997.tb12595.x — Morschhäuser, Joachim, et al. "Degradation of human subendothelial extracellular matrix by proteinase-secreting Candida albicans." FEMS microbiology letters 153.2 (1997): 349-355.
- http://dx.doi.org/10.1080/13693780410001657162 — Dolan, Joseph W., et al. "Candida albicans PLD1 activity is required for full virulence." Medical mycology 42.5 (2004): 439-447. PDF Available Here
- http://dx.doi.org/10.1086/319599 — Sebti, Abdelghani, et al. "Candida dubliniensis at a cancer center." Clinical infectious diseases 32.7 (2001): 1034-1038.
- http://dx.doi.org/10.1128%2FJCM.01113-08 — Sheppard, Donald C., et al. "Utility of the germ tube test for direct identification of Candida albicans from positive blood culture bottles." Journal of clinical microbiology 46.10 (2008): 3508-3509. Full Text Available Here
- http://www.ncbi.nlm.nih.gov/pubmed/9145613 — Prakash, Prabhu, Aruna Solanki, and K. R. Joshi. "A simple synthetic liquid medium for development of yeast and mycelial form of pathogenic species of Candida." Indian Journal of Pathology and Microbiology 40 (1997): 55-58.
- https://doi.org/10.1128/CMR.12.1.80 -- Fidel, Paul L., Jose A. Vazquez, and Jack D. Sobel. "Candida glabrata: review of epidemiology, pathogenesis, and clinical disease with comparison to C. albicans." Clinical Microbiology Reviews [12.1 (1999): 80-96].
- https://pubmed.ncbi.nlm.nih.gov/22041440/ -- Lin XL, Li Z, Zuo XL. "Study on the relationship between vaginal and intestinal candida in patients with vulvovaginal candidiasis." Chinese Journal of Obstetrics and Gynecology (Zhonghua fu chan ke za zhi). [2011 Jul;46(7):496].